 Estimation errors arising from variations in the quantification method can be the result of DNA integrity, enzyme efficiency and many other factors. Although this technique is still used to assess gene expression, it requires relatively large amounts of RNA and provides only qualitative or semi quantitative information of mRNA levels. In this method, purified RNA is separated by agarose gel electrophoresis, transferred to a solid matrix (such as a nylon membrane), and probed with a specific DNA or RNA probe that is complementary to the gene of interest. Northern blotting is often used to estimate the expression level of a gene by visualizing the abundance of its mRNA transcript in a sample. Older methods were used to measure mRNA abundance: differential display, RNase protection assay and northern blot. Quantitative PCR and DNA microarray are modern methodologies for studying gene expression. This measurement is made after each amplification cycle, and this is the reason why this method is called real time PCR (that is, immediate or simultaneous PCR). Quantitative PCR can also be applied to the detection and quantification of DNA in samples to determine the presence and abundance of a particular DNA sequence in these samples. The data thus generated can be analysed by computer software to calculate relative gene expression (or mRNA copy number) in several samples. This allows the rate of generation of the amplified product to be measured at each PCR cycle. A substance marked with a fluorophore is added to this mixture in a thermal cycler that contains sensors for measuring the fluorescence of the fluorophore after it has been excited at the required wavelength allowing the generation rate to be measured for one or more specific products. In order to amplify small amounts of DNA, the same methodology is used as in conventional PCR using a DNA template, at least one pair of specific primers, deoxyribonucleotide triphosphates, a suitable buffer solution and a thermo-stable DNA polymerase. The polymerase chain reaction (PCR) is a common method for amplifying DNA for RNA-based PCR the RNA sample is first reverse-transcribed to complementary DNA (cDNA) with reverse transcriptase. In order to robustly detect and quantify gene expression from small amounts of RNA, amplification of the gene transcript is necessary. Real time PCR uses fluorophores in order to detect levels of gene expression.Ĭells in all organisms regulate gene expression by turnover of gene transcripts (single stranded RNA): The amount of an expressed gene in a cell can be measured by the number of copies of an RNA transcript of that gene present in a sample. The acronym "RT-PCR" commonly denotes reverse transcription polymerase chain reaction and not real-time PCR, but not all authors adhere to this convention. The Minimum Information for Publication of Quantitative Real-Time PCR Experiments ( MIQE) guidelines propose that the abbreviation qPCR be used for quantitative real-time PCR and that RT-qPCR be used for reverse transcription–qPCR. Two common methods for the detection of PCR products in real-time PCR are (1) non-specific fluorescent dyes that intercalate with any double-stranded DNA and (2) sequence-specific DNA probes consisting of oligonucleotides that are labelled with a fluorescent reporter, which permits detection only after hybridization of the probe with its complementary sequence. Real-time PCR can be used quantitatively and semi-quantitatively (i.e., above/below a certain amount of DNA molecules). It monitors the amplification of a targeted DNA molecule during the PCR (i.e., in real time), not at its end, as in conventional PCR. 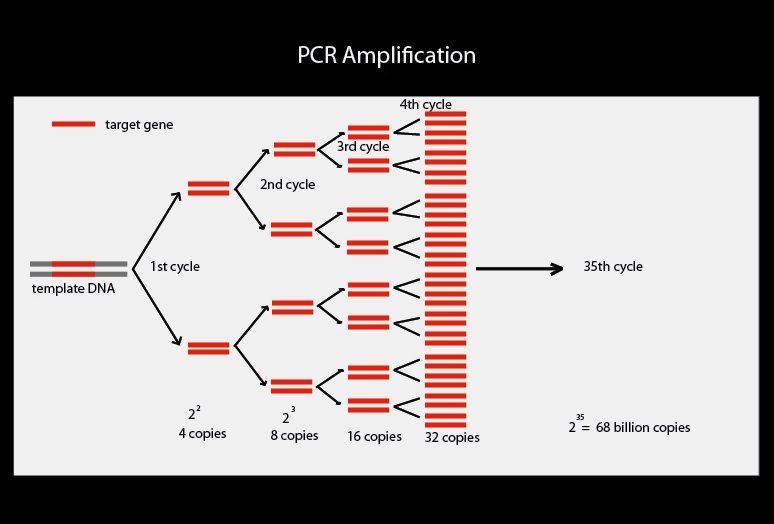
Melting curve produced at the end of real-time PCRĪ real-time polymerase chain reaction ( real-time PCR, or qPCR when used quantitatively) is a laboratory technique of molecular biology based on the polymerase chain reaction (PCR). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
